Nigeria grants approval for Scottish life science firm's HIV test
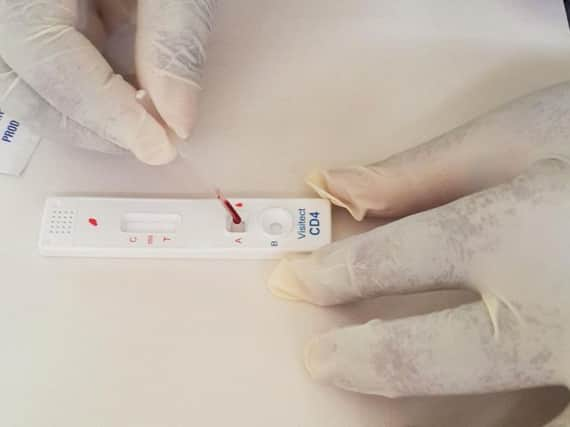

The group, which is based in Alva, Clackmannanshire, and specialises in tests for allergies, infectious diseases and food intolerance, said it had received approval from the Federal Ministry of Health to enable the Visitect CD4 test to be deployed in Nigeria.
The test will be adopted into the country’s national HIV control programme as a “rapid and instrument-free test device that provides actionable results at the point of care”, Omega added.
Last month, the group said it had received initial orders from its distribution partner in Nigeria for 250,000 tests. It will now look to “firm up” a delivery schedule to determine what proportion of this demand can be shipped prior to the end of the financial year.
Chief executive Colin King said: “I am pleased that our test has been approved for use in Nigeria which was one of our key strategic aims and allows our unique point-of-care test to make a real difference to people living with HIV in Nigeria.”