Scots life sciences firm Omega upbeat on Covid self-test kit progress

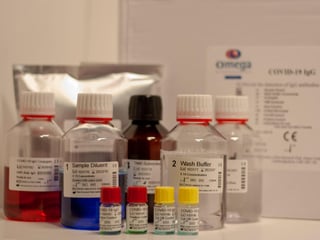
A consortium which is led by Abingdon Health and that includes Omega, which is based in Alva, Clackmannanshire, is working with the Medicines and Healthcare products Regulatory Agency (MHRA) to approve the lateral flow antibody test.
In its update, Abingdon said that while it continued to liaise with the MHRA regarding approval for home use and self-test of the AbC-19 Rapid Test for antibodies, it was liaising with regulatory authorities in other jurisdictions around the world to allow the use of the product.
Advertisement
Hide AdAdvertisement
Hide AdColin King, chief executive of Omega Diagnostics, said: “We welcome the update from Abingdon Health and we look forward to being able to update investors on this approval process for self-test use in due course.
“We remain confident that the AbC-19 Rapid Test has a potential global application and we are delighted that the UK-RTC maintains constructive dialogue with interested customers and regulatory authorities across the world.”
At the end of November, Omega reported a 29 per cent fall in first-half revenue to £3.16 million. Gross margin decreased to 42.9 per cent from 67.5 per cent a year earlier, reflecting the fixed nature of labour costs on reduced sales.
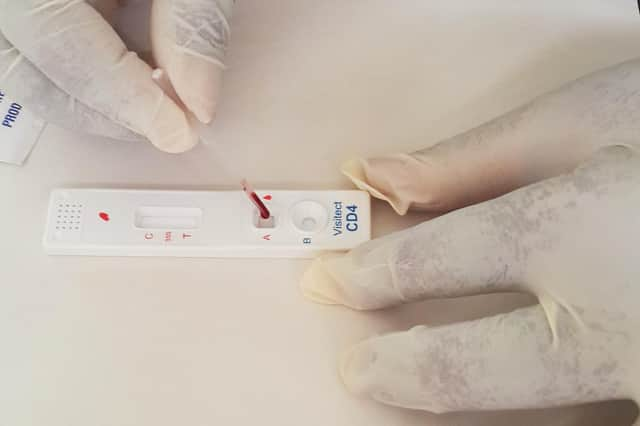
However, bosses pointed to “substantial” growth opportunities in the areas of food intolerance, CD4 testing for the management of people living with HIV, and Covid-19 antibody and antigen testing. The firm provides in-vitro diagnostics products for use in hospitals, clinics, laboratories and healthcare practices in some 75 countries.
A message from the Editor:
Thank you for reading this article. We’re more reliant on your support than ever as the shift in consumer habits brought about by coronavirus impacts our advertisers. If you haven’t already, please consider supporting our trusted, fact-checked journalism by taking out a digital subscription: www.scotsman.com/subscriptions
Comments
Want to join the conversation? Please or to comment on this article.