UK set to pioneer use of human embryo gene editing
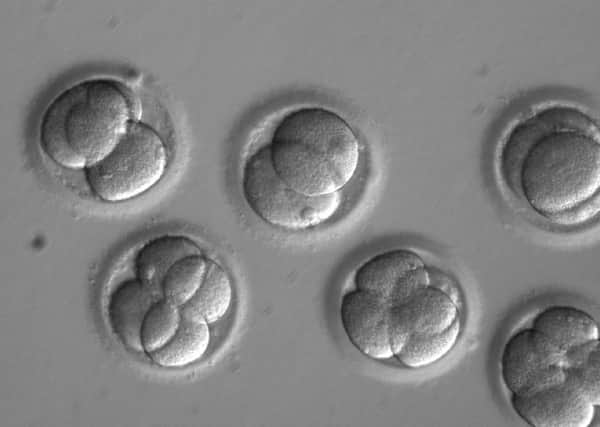

The landmark study, first revealed in The Scotsman’s sister paper the i last week, came after scientists in the US reached a milestone by successfully altering DNA in defective embryos so they were no longer programmed to develop heart failure.
It is the first time the powerful gene-editing tool Crispr-Cas9 has been used to fix a mutation responsible for a common inherited disease.
Advertisement
Hide AdAdvertisement
Hide AdThe highly controversial technique is still at an early experimental stage and there is no question of any attempt being made to create babies with the genetic modification, which would be illegal both in the US and the UK.
But a leading member of the team has hinted that first steps towards bringing the treatment to patients could take place in the UK under the direction of the fertility regulator the Human Fertilisation and Embryology Authority (HFEA).
Dr Shoukhrat Mitalipov, from Oregon Health and Science University (OHSU) in Portland, said: “Maybe… [the] HFEA might take a lead on this, but I’m quite sure before these clinical trials can go on they have to go through, I believe, Parliament to change a law. So there is still a long road ahead, particularly if you want to do it in a regulatory way.”
US regulatory barriers to such research are so high they could be insurmountable. In the US, taxpayer funds cannot pay for research that destroys human embryos.
And Congress has banned the US Food and Drug Administration (FDA) from even considering the possibility of human clinical trials involving embryos with edited inherited genes.
More liberal Britain has already blazed a trail by becoming the first country officially to sanction mitochondrial replacement therapy (MRT), seen by some as opening the door to “designer babies”. The treatment replaces faulty inherited DNA in the mitochondria, tiny rod-like bodies in cells that supply energy, to prevent devastating diseases.
However, the new research, reported in the journal Nature, goes much further by modifying the nuclear DNA at the heart of the cell that influences personal characteristics such as height, facial appearance, eye colour and intelligence.
Professor Peter Braude, from King’s College London, said: “With this paper the possibility of germline genome editing has moved from future fantasy to the world of possibility, and the debate about its use, outside of fears about the safety of the technology, needs to run to catch up.”
Advertisement
Hide AdAdvertisement
Hide AdStem cell scientist Professor Robin Lovell-Badge, group leader at the Francis Crick Institute in London, stressed that the research was “not about designer babies”.
He said: “There is still much to be done to establish the safety of the methods, therefore they should not be adopted clinically.
“However, this method of genome editing could, in theory, be harnessed to correct mutations coming from a father.”